Research Areas
Protein Dynamics in Autoimmunity
Nanoparticles and Immunogenicity
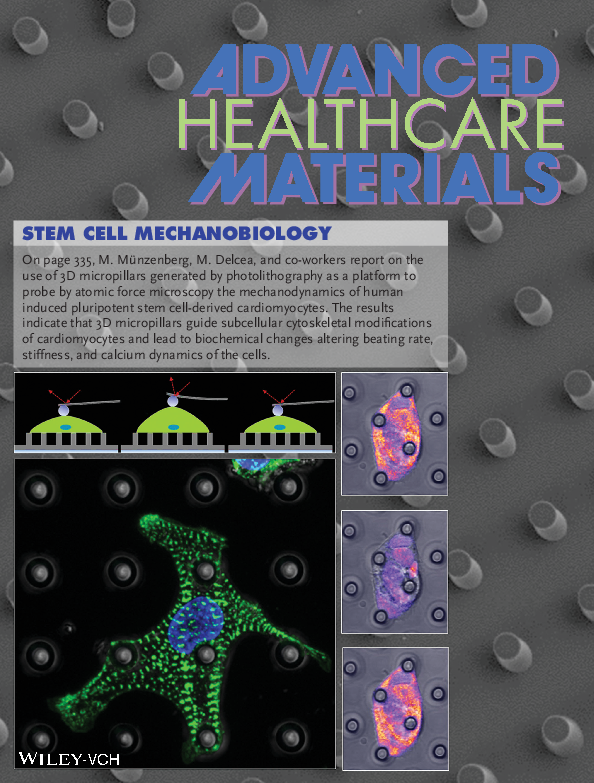
Mechanobiology of iPSC-derived cardiomyocytes
1. Protein Dynamics in Autoimmunity
A major challenge in the study of protein dynamics is the visualization of changing conformations that are important for processes ranging from enzyme catalysis to signaling. In addition, protein dynamics studies are relevant for investigating the equilibrium of protein conformations causing diseases
We combine biophysical techniques (e.g. circular dichroismus spectroscopy, single molecule spectroscopy, calorimetry, fluorescence microscopy, atomic force microscopy) and molecular dynamics simulation studies to investigate the impact of mutations, posttranslational modifications or stress conditions (e.g. pH, drugs) on the protein dynamics.
Of particular interest is the conformational dynamics of proteins involved in autoimmune diseases (e.g. soluble beta2-glycoprotein and transmembrane protein alpha2b beta3).
2. Nanoparticles and Immunogenicity

We investigate several aspects related to nanoparticles (NPs). We focus on the synthesis, functionalization and characterization of NPs that are used as specific intracellular probes, tumor visualization and localized photothermal treatment, etc.
NPs can stimulate and/or suppress the immune response by binding to blood proteins. Although the compatibility of the NPs with the immune system is largely determined by their physico-chemical properties (e.g. size, shape, charge, surface groups), the effect of nanoparticle properties on the immune system is little explored. Using in vitro tools we predict and control the immunogenicity of NPs by adjusting their physical properties (e.g. size, surface charge) and targeting ligands (e.g. antibodies, proteins).
We also investigate the interaction of plastic NPs with proteins and cells using biophysical methods (e.g. calorimetry, dynamic light scattering, quartz crystal microbalance, microscopy, etc) towards development of methods to detect plastic NPs in the body.
3. Mechanobiology of iPSC-derived cardiomyocytes
Induced pluripotent stemm cell derived cardiomyocytes (iPSC-CM) possess outstanding capacity for cardiac regeneration and represent models of cardiac diseases.
We investigate biochemical changes and intracellular reorganization of the sarcomeres which mediate signaling and beating rate of iPSC-CM. Three-dimensional (3D) micropillars generated by photolithography and functionalized with biopolymers which mimic the extracellular matrix of structural proteins and other biological molecules. Such 3D substrates are used to study mechanobiology of patient-specific induced pluripotent stem cells (e.g. cell elasticity, contractility and organization of the cytoskeleton). In addition, molecular changes between healthy and diseased samples, expression of cardiomyocytes maturation markers is investigated.
Prof. Ali Abou-Hassan (Sorbonne University, Paris, France)
Prof. Adnane Achour (Karolinska Institute, Stockholm, Sweden)
Prof. Christiane A. Helm (Institute of Physics, University Greifswald, Germany)
Prof. Markus Münzenberg (Institute of Physics, University of Greifswald, Germany)
Prof. Oliver Otto (Institute of Physics, University of Greifswald, Germany)
Prof. Christian Schmidt (Internal Medicine C, University of Greifswald, Germany)
Prof. Jose Luis Toca-Herrera (Institute of Biophysics, University of Natural Resources and Life Sciences Vienna, Austria)
Prof. Karen Vanhoorelbeke (KU Leuven, Belgium)
Prof. Thomas McDonnell (University College London, United Kingdom)
Prof. Liraz Chai (The Hebrew University of Jerusalem, Israel)
- Prof. Andreas Kupz (Australian Institute of Tropical Health and Medicine, James Cook University, Australia)
- Prof. Mikael Björnstedt (Karolinska Institute, Stockholm, Sweden)
aktualisiert am 06.05.2026 durch